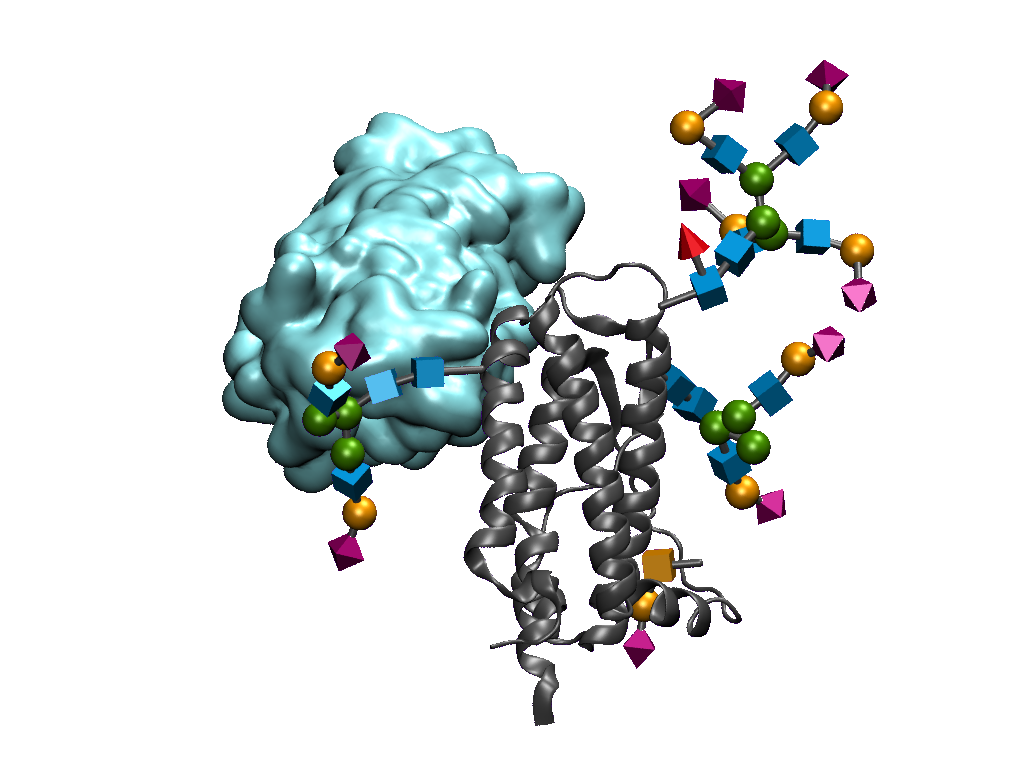
To investigate the differences between reagents used to analyze N-glycans, we benchmarked our N-GlyFind™ Core-Specific Reagent against three commonly used lectins using the same 32 N-glycan array from Gao et al. (2019), allowing for a direct comparison of structural recognition across the four carbohydrate-binding proteins.
Concanavalin A (ConA)
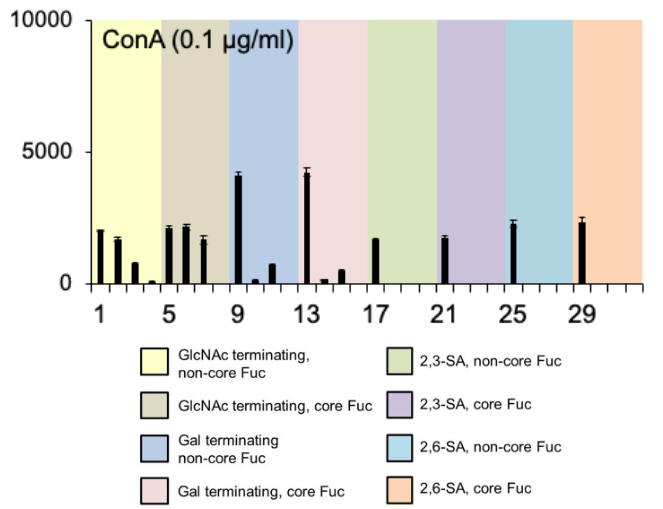
ConA bound only biantennary N-glycans, and weakly to GlcNAc-terminating triantennary species
Adapted from Gao et al. (2019)
L-PHA
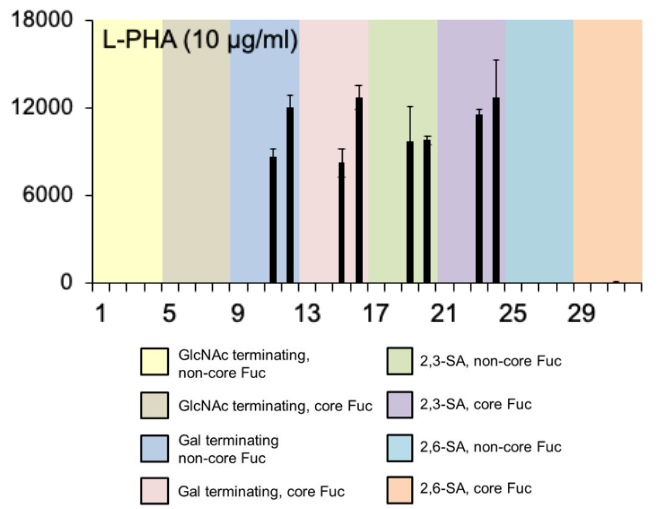
L-PHA demonstrated a restricted binding pattern, only binding to the β1,6 branch of N-glycans in the presence of non-α2,6-sialylated LacNAc structures
Adapted from Gao et al. (2019)
E-PHA
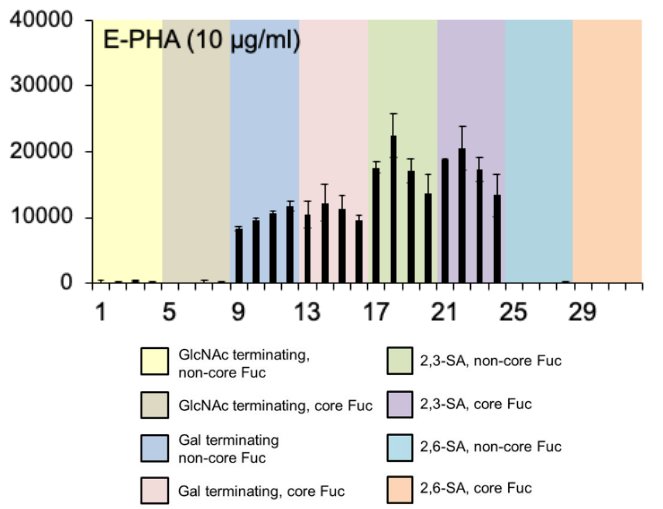
The E-PHA binding pattern included bi-, tri-, and tetraantennary N-glycans with or without α2,3-linked sialic acids, but no binding to GlcNAc-terminating or α2,6-sialylated N-glycans
Adapted from Gao et al. (2019)
N-GlyFind™ Core-Specific Reagent
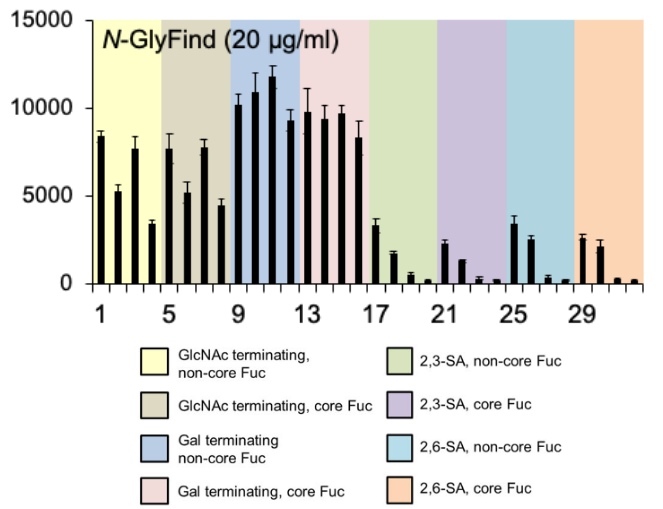
Compared to the previous lectins, N-GlyFind™ bound a wider array of common N-glycan structures
Lectenz Bio and NCFG, unpublished